If you hand a neuropharmacologist three unlabeled compounds — methylphenidate, amphetamine, and cocaine — and ask what they do, the answer will involve the same short list of neurotransmitters, the same receptors, and the same brain regions. If you hand the same three compounds to a judge, the answer will involve three completely different legal categories. One of these people is looking at the chemistry. The other is looking at the century.
This article is about the chemistry. Specifically, it is about why the dividing line between "legitimate ADHD medication" and "Schedule I contraband" has very little to do with what these molecules do inside a brain, and almost everything to do with how fast they get in and how fast they leave.
Once you understand that — once you see pharmacokinetics as the actual variable rather than pharmacology — the entire legal architecture around coca starts to look less like science and more like a historical accident that nobody has bothered to revisit.
What a stimulant actually does
To follow the argument, you need one concept: the monoamine system. Your brain uses several small-molecule messengers to regulate attention, motivation, alertness, and mood. The three most relevant ones for this discussion are dopamine, norepinephrine, and serotonin. Collectively, they are called monoamines.
After a neuron releases one of these molecules into the gap between itself and the next neuron, specialized transporter proteins — DAT (dopamine transporter), NET (norepinephrine transporter), and SERT (serotonin transporter) — pull the molecules back into the releasing cell so they can be used again. This is called reuptake, and it is how the brain regulates how much signal is available in the synapse at any given moment.
Every classical psychostimulant interferes with this process. They differ mainly in which transporters they target and how they interfere.
Methylphenidate (Ritalin, Concerta, Focalin)
Methylphenidate is a reuptake inhibitor. It binds to DAT and NET and prevents them from clearing dopamine and norepinephrine out of the synapse. The result is increased availability of both neurotransmitters in the brain regions involved in attention and executive function. It has little direct action on serotonin.
Amphetamine (Adderall, Vyvanse, Dexedrine)
Amphetamine does something slightly different. It is both a reuptake inhibitor and a releasing agent: it actively causes neurons to dump stored dopamine and norepinephrine out into the synapse. This is a more aggressive mechanism than simple reuptake inhibition. It produces a larger, more sustained elevation in monoamine signaling — which is one reason amphetamines generally have a stronger subjective effect than methylphenidate at equivalent doses.
Cocaine
Cocaine is a reuptake inhibitor — like methylphenidate — but with a broader footprint. It blocks DAT, NET, and SERT. That means it increases availability of dopamine, norepinephrine, and serotonin simultaneously. Mechanistically, this places cocaine in the same general category as methylphenidate, with an additional serotonergic effect.
Three drugs. Three slightly different handles on the same basic system. This is not a controversial description; it is what every pharmacology textbook says.
Look at the molecules
There is a visual version of this argument that moves faster than any amount of prose. Methylphenidate and cocaine are, structurally, built from the same small set of chemical parts. If you line them up and color the shared features, the relationship stops being an analogy and starts being a fact.
That green ring — the piperidine — is where both molecules anchor their grip on the transporter proteins that clear monoamines out of the synapse. The gold ring is the phenyl, a hydrophobic handle that drops into a lipophilic pocket of the transporter. The blue fragment is the methyl ester that both compounds carry as their business end. Three pieces, in roughly the same arrangement, each doing roughly the same job. This is what a monoamine reuptake inhibitor looks like when you strip it down to its functional parts.
Cocaine has more scaffolding around those parts. The benzoate ester attaches the phenyl one carbon further out from the piperidine than methylphenidate's direct connection. The ethylene bridge across the nitrogen turns the simple piperidine into the bicyclic tropane system that is the hallmark of the natural-product alkaloid family. These additions matter for stereochemistry, for metabolism, and for exactly how tightly the molecule binds its targets. What they do not change is the fundamental category. A medicinal chemist looking at these two drawings sees the same type of molecule twice — a pair of close cousins separated by a couple of evolutionary decorations the coca plant happened to add and the Swiss chemists who synthesized methylphenidate happened to leave off.
Skeletal formulas are a simplification. Here are the same two compounds rendered in three dimensions, atoms drawn to their real relative sizes:
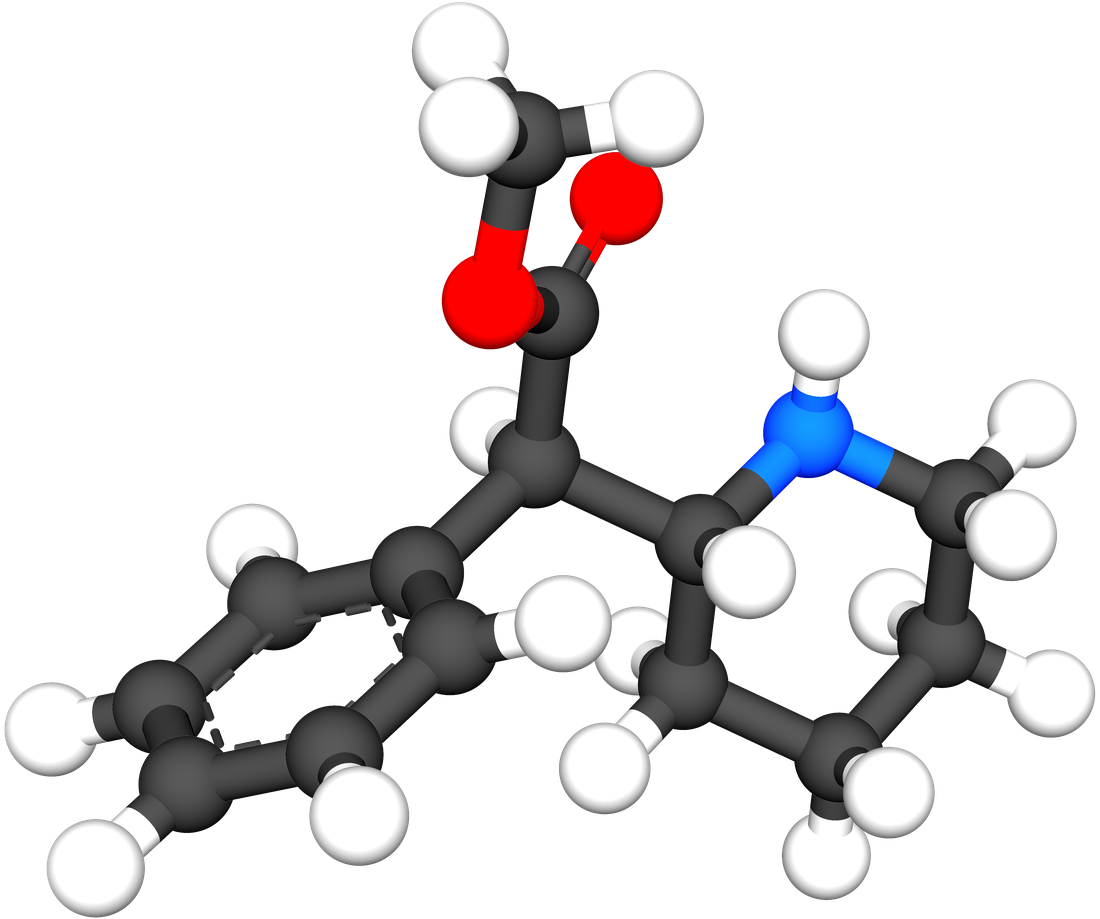
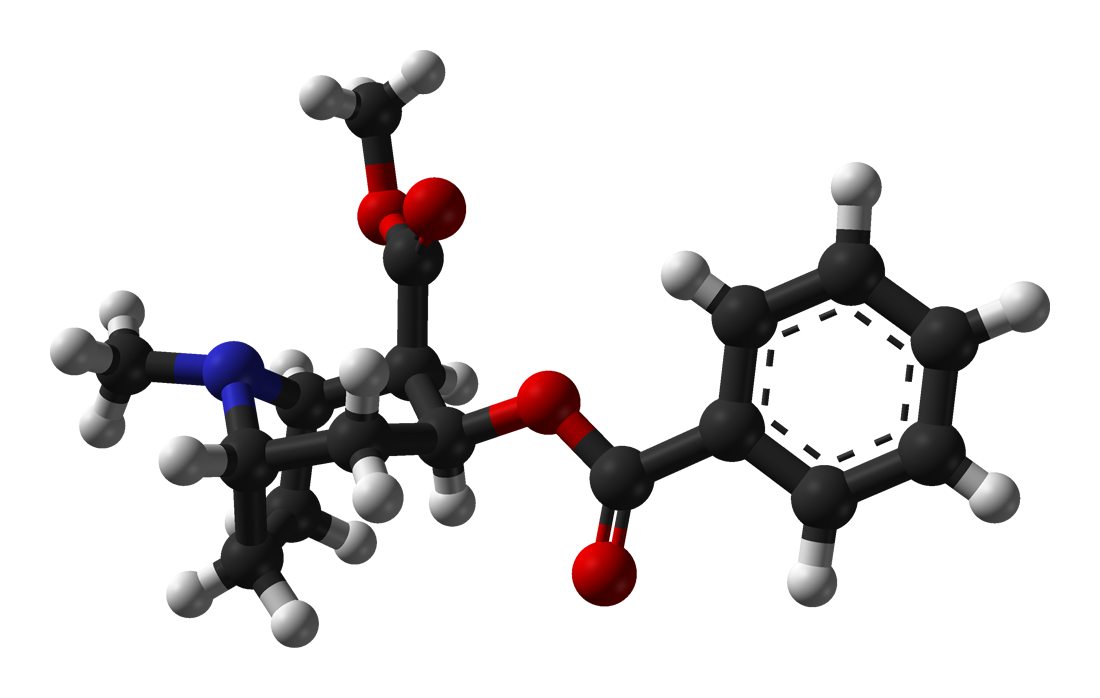
Both objects are roughly the same size. Both carry the same three functional landmarks — a saturated six-ring with a nitrogen (the piperidine, on the right of methylphenidate and on the left of cocaine), an aromatic benzene ring, and a methyl ester with its two characteristic oxygen atoms. The extra structure on cocaine — the second ester linker, the ethylene bridge arching across the nitrogen — is real, and it is not trivial, but it is not what the molecule is. It is what the molecule wears. Strip it off and what's underneath is something a pharmacologist would immediately recognize as belonging to the same family as the pill the kid in seat 3B took at breakfast.
Methylphenidate and cocaine are, at the level of mechanism, closer to each other than either is to amphetamine. And yet methylphenidate is a lunchbox medication and cocaine is a life sentence. The mechanism nobody wants to say out loud
The table nobody publishes
Here is what this actually looks like when you put it side by side. These values are generalizations — exact pharmacokinetic numbers vary with formulation, route, and individual — but the shape of the comparison is stable across the research literature.
Look at that last row carefully. Methylphenidate, amphetamine, and the coca leaf are all Schedule II substances under U.S. federal law. Legally, they are classified at the same level. The difference is entirely in how that classification plays out in practice. Two of them are routinely prescribed to American children. The third is functionally impossible to obtain because the FDA has never approved any coca-leaf product and no manufacturer can legally produce one.
The law is not treating these compounds according to their pharmacological danger. It is treating them according to their cultural history.
The variable that actually matters: pharmacokinetics
If the mechanisms are broadly comparable, where does the genuine difference between a medicine and an addiction come from? The answer is not what the compound does, but how quickly it does it and how quickly it stops.
Pharmacokinetics describes the concentration curve of a drug in the bloodstream and, by extension, at the site of action in the brain. There are two numbers that dominate everything else:
- Tmax — the time from dosing to peak concentration. A short Tmax means the drug hits hard and fast.
- Half-life — how long the drug takes to clear. A short half-life means the drug leaves almost as fast as it arrived.
Addiction research has consistently shown that the rate of onset — the slope of that rising curve — is one of the strongest predictors of whether a compound will produce compulsive use. Slow-onset drugs teach the brain a relationship between behavior and gradual mood change. Fast-onset drugs teach the brain that a single action produces an immediate, enormous reward. The second pattern is the one that hijacks the reinforcement system.
Look at that shape. The red curve — insufflated cocaine hydrochloride — rises almost vertically, peaks, and then falls away sharply. That fast rise-fall signature is what the addiction literature calls reinforcement-efficient delivery. It is also exactly the signature that any responsible pharmacology would want to avoid in a therapeutic product.
The green curve — slow-release methylphenidate — is almost the opposite. It rises gradually, plateaus for hours, and declines smoothly. This shape is deliberately engineered. When methylphenidate was first sold as Ritalin in an immediate-release tablet, it had a higher abuse potential than the extended-release formulations that followed. Pharmaceutical companies learned — and the FDA required — that flattening the curve made the drug safer. Concerta, Focalin XR, Vyvanse (which is a prodrug of amphetamine designed to release slowly) all represent the same principle: slow the onset and you keep the therapeutic effect while cutting the abuse liability.
And then there is the gold curve in the middle. That is the coca leaf as it is traditionally used — chewed slowly with an alkaline activator, releasing alkaloids gradually through the oral mucosa over a period of many minutes. The shape of that curve looks much more like the slow-release prescription stimulant than it does like the refined cocaine signature at the top of the chart.
This is not a coincidence. It is the reason Andean cultures could integrate coca into eight thousand years of daily life without producing the kind of addiction epidemic that cocaine produced almost immediately after it was isolated. The plant was already a slow-release formulation. Nobody had to engineer it that way. Evolution and traditional preparation got there first.
Pharmaceutical companies spend enormous resources reformulating existing stimulants to slow their onset and reduce abuse potential. The coca leaf, prepared in the way Andean people have prepared it for millennia, already has the pharmacokinetic profile those reformulations are trying to achieve. The engineering work has already been done. It was done by the plant.
What this means for ADHD
ADHD treatment today is dominated by two classes of drugs — methylphenidate derivatives and amphetamine derivatives — along with a small number of non-stimulant alternatives like atomoxetine and guanfacine. Between them, stimulant prescriptions account for the vast majority of pharmacological ADHD treatment in the United States, Canada, Europe, and the wealthier parts of Latin America.
These drugs work. The evidence base is substantial. But they come with a well-known cluster of side effects: insomnia, appetite suppression, elevated heart rate and blood pressure, emotional blunting at higher doses, rebound irritability when the dose wears off, and — in some patients — an uncomfortable subjective feeling that is often described as being "wired" or "not myself." Some patients tolerate these effects comfortably. Others do not, and spend years cycling through dose adjustments and drug switches looking for a formulation their body agrees with.
A standardized, slow-release coca-leaf extract is, pharmacologically, a plausible candidate in this space. Here is why:
- Mechanism overlap. Monoamine reuptake inhibition is exactly what current ADHD stimulants target. The mechanism is not speculative.
- Favorable kinetics. The leaf's slow buccal absorption produces the low-peak, moderate-duration curve that modern ADHD formulations are specifically designed to mimic.
- Buffering by the full alkaloid spectrum. Coca leaf contains more than a dozen alkaloids in addition to the principal one. Whole-plant extracts preserve this matrix, which appears to moderate the subjective intensity in a way the isolated alkaloid does not.
- A long safety record at the leaf level. Traditional use patterns provide an observational dataset that is larger, longer, and more culturally embedded than the entire existence of the ADHD diagnosis.
None of this means the leaf is a ready-to-ship ADHD therapeutic. It means it is a ready-to-study ADHD therapeutic. There is a meaningful difference, and the difference is a clinical trial program — which is precisely what the current scheduling framework makes almost impossible to run.
The question the pharmacology keeps asking
If the dividing line between a medicine and a felony is delivery speed, then the legitimate regulatory question isn't "should coca be legal?" It's "which preparations of coca have pharmacokinetic profiles that belong in a medical framework, and which don't?"
That is a question you can actually answer with research. It has clean inputs, measurable outputs, and a clear decision rule: anything with an onset curve that resembles insufflated cocaine hydrochloride belongs under restriction, and anything with an onset curve that resembles extended-release methylphenidate belongs under the same framework we already use for extended-release methylphenidate. The chemistry is the same. The categories should follow the chemistry.
We already make exactly this distinction for opioids. Nobody confuses oxycodone extended-release with heroin, even though they are closely related molecules that hit overlapping receptors. We distinguish them by formulation, by delivery route, by dose, and by medical context. The entire modern opioid regulatory apparatus — for all its well-documented failures — at least recognizes that the molecule and the preparation are different things that deserve different treatment.
The coca leaf has not yet been granted that distinction. The pharmacology says it should be.
None of what is written here is new. Every claim in this article is consistent with published pharmacology, with standard textbook descriptions of monoamine reuptake mechanisms, and with the pharmacokinetic principles that already govern modern ADHD drug development. What is new is simply putting the three compounds on the same page and letting the comparison speak.
When you do that, the current legal framework stops looking like science. It starts looking like an artifact — a set of rules written before the pharmacokinetic data existed, carried forward by institutional momentum, and never revisited because nobody with political capital wanted to be the person who revisited it.
Somebody is going to revisit it eventually. The question is whether that revisiting happens in a research hospital in Bogotá, in a pharmaceutical lab in Lima, in a clinical trial in La Paz — or whether the countries that know this plant best get left out of the conversation once again.